Tilo Schmutzler
Tilo Schmutzler, Dr. rer. nat.
Project description
Gold nanoparticles (AuNPs) exhibit outstanding optical properties due to their plasmonic behavior. The absorption of UV- visible or NIR-light can be tuned by controlling size and shape of the AuNPs.[1] Gold nanorods (AuNRs) for example show two absorption bands due to their anisotropic shape.[2] To modify the morphology of AuNPs, surface-active substances like surfactants are needed to control formation, growth and stabilization of such particles. Cetyltrimethylammonium bromide (CTAB) is one of the most common surfactants and is used to prepare AuNRs in high yields and low polydispersity.[3] Yield and polydispersity can be influenced by various additives (Fig.1).
It is known, that CTAB forms micelles in aqueous solutions and a double layer to stabilize AuNRs.[4] We found that the stabilization of especially small AuNPs needs additional sterical stabilization which is achieved using high CTAB concentrations and thus a high amount of micelles in solution. The interaction between AuNPs and CTAB micelles can be complex but might be one of the major influences during AuNP formation and stabilization.
To characterize both AuNPs and CTAB micelles we use small angle scattering of X-ray (SAXS) and neutrons (SANS). The purpose of this project is to understand the interactions between AuNPs and micelles combining in situ SAXS and ex situ SAXS and SANS complemented with UV-Vis-NIR spectroscopy and transmission electron microscopy (TEM) (Fig.2). The addition of KBr for example influences both the AuNR formation and the structure of CTAB micelles.
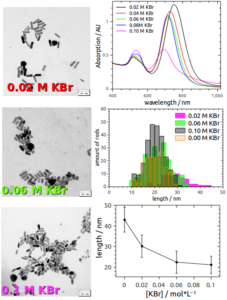
Fig.1: TEM and UV-Vis-NIR spectroscopy can be used to characterize gold nanorods synthesized under various KBr concentrations.
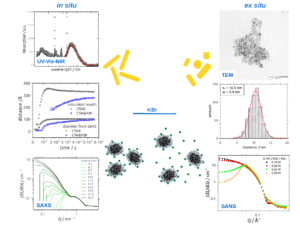
Fig.2: Combining simultaneous in situ UV-Vis-NIR spectroscopy and SAXS with ex situ TEM and SANS leads to an deep understanding of the interaction between gold nanorods and CTAB micelles.
References
[1] J. Pérez-Juste et al., Coord. Chem. Rev. 249 (2005) 1870.
[2] C.J. Orendorff et al., Small 2 (2006) 636.
[3] N.R. Jana et al., Adv. Mater. 13 (2001) 1389.
[4] S. Gómez-Grana et al., Langmuir 28 (2012) 1453.

