Moses Richter
Moses Richter, M.Sc.
Project description
Solution cast thin films of functional nanoparticles are important to many novel printed electronics, e.g. organic solar cells, organic LEDs and OFETs. For instance, ZnO nanoparticles are often utilized as selective electron transport layer, due to its abundance and environmental benefits.[1] However, a reproducible and stable ink formulation requires the use of stabilizing ligands, which tend to strongly influence the electrical conduction in the dried film and thus deteriorate the device performance. In this project ZnO and Al doped ZnO (AZO) nanoparticle films are investigated towards their bulk-conductivity in as-cast films, to gain better inside into limiting factors. The electric measurements are conducted on thick films sandwiched between an ITO and Al electrode, as illustrated in figure 1, inside a N2-flow cryostat for temperature and atmospheric control. Eventually, the electric transport is determined as Poole-Frenkel limited conduction (see figure 2) with trap-barriers around 100 meV.[2] The use of ligands causes the intrinsic trap-barrier either to increase or decrease, however the amount of ligands outweighs this effect and reduces the overall conductivity even further. In order to achieve full performance in e.g. organic solar cells incorporating a ZnO interlayer, one commonly applies a UV-soaking step after device fabrication. UV light is known to cause oxygen desorption from the ZnO surface, releasing trapped electrons which in excess make the material conductive and ohmic, respectively.[3,4] Along with the change in charge transport the conductivity also increases by up to 3 orders of magnitude. The effect is persistent under oxygen exclusion, however, the activation is not maintained in solar cell devices. Processing our own devices, we applied the UV-soaking step right before subsequent layers were cast onto the film of AZO nanoparticles in order to release adsorbed oxygen and thereby stabilized the solar cell performance for a much longer time period.
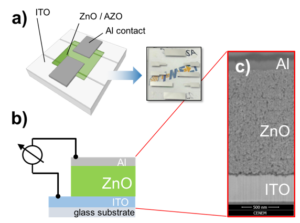
Figure 1: a) The bulk-conductivity in ZnO and AZO nanoparticles films is characterized in “sandwich” devices. b) illustrate the cross section and c) shows the SEM micrograph through a 1.5µm thick film of ZnO.
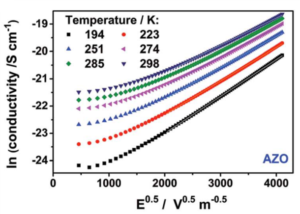
Figure 2: The bulk conduction of a Al doped ZnO nanoparticle film is limited by an energetic trap leading to a Poole-Frenkel transport, as indicated by the linear dependency in this Poole-Frenkel plot.[2]
References
[1] T. Stubhan et al., Org. Electron. 12 (2011) 1539.
[2] T. Lenz et al., J. Mater. Chem. C 3 (2015) 1468.
[3] A. J. Morfa et al., Sol. Energy Mater. Sol. Cells 124 (2014) 211.
[4] S. Wilken et al., J. Phys. Chem. C 118 (2014) 19672.

